Refractory chronic patellar tendinopathy is one of the most disabling injuries of the musculoskeletal system, particularly in athletes and active patients. When conservative treatments fail to provide a lasting solution, many patients explore biological alternatives: PRP, injections of non-expanded cells, or preparations marketed under labels such as “stem cells” or “regenerative therapies” (unregulated terms that are not classified as medicinal products and do not require AEMPS authorization).
An advanced cell therapy medicinal product based on cultured mesenchymal cells (CMC) belongs to a different category: GMP cleanroom manufacturing, dose validated in clinical trial, and a specific regulatory framework under AEMPS. Below is a summary of what the available clinical evidence shows.
What is chronic patellar tendinopathy and why is it so difficult to treat?
The patellar tendon connects the kneecap to the tibial tuberosity. In chronic tendinopathy, the tissue undergoes progressive degeneration that, in the most severe cases, evolves into a structural tear with fiber separation visible on MRI. Unlike acute tendinitis, it does not respond well to rest or conventional anti-inflammatory drugs, and when conservative treatment fails (physical therapy, shockwave therapy, injections), the usual alternative is surgery.
Degenerated tendon tissue has limited self-regeneration capacity and requires interventions that act on the biology of the tendon itself, not solely on pain.
See also: Clinical advances in patellar tendinopathy with Cultured Mesenchymal Cells (CMC)
What are CMC and how do they differ from other biological treatments?
In recent years, a number of procedures have been promoted as regenerative alternatives: PRP, secretomes, PRP-hexosome, or stromal vascular fraction. They all share a common pattern: they promise regeneration without expanded living cells, without a validated dose, and without a regulatory framework as a medicinal product.
The cultured mesenchymal cells (CMC) are an advanced cell therapy medicinal product obtained from the patient’s own bone marrow. They are isolated, expanded in a GMP cleanroom, and administered at a clinically validated dose within the regulatory framework of the AEMPS. They are not a platelet concentrate: they are living cells capable of secreting trophic factors, modulating inflammation, and stimulating structural regeneration of the damaged tendon.
See also: Cell dose in cultured mesenchymal cells (CMC): the parameter that changes everything
The comparative clinical trial: CMC versus PRP in patellar tendinopathy
ITRT sponsored a comparative clinical study in patients with chronic patellar tendinopathy who had not responded to conventional treatments and presented a fiber tear visible on MRI (≥3 mm). It was a clinical trial of the highest scientific rigor: with random assignment, neither the patient nor the physician knew which treatment each person was receiving (double-blind), and outcomes were objectively compared between the two groups.
The study involved researchers from ITRT, the FC Barcelona Medical Services, the University of Valladolid, and the Centro Médico Teknon, among other institutions. The results were published in two leading international scientific journals in sports medicine: first the 6-month results in The American Journal of Sports Medicine (2021), and later the 12-month follow-up results in the Orthopaedic Journal of Sports Medicine (2023).
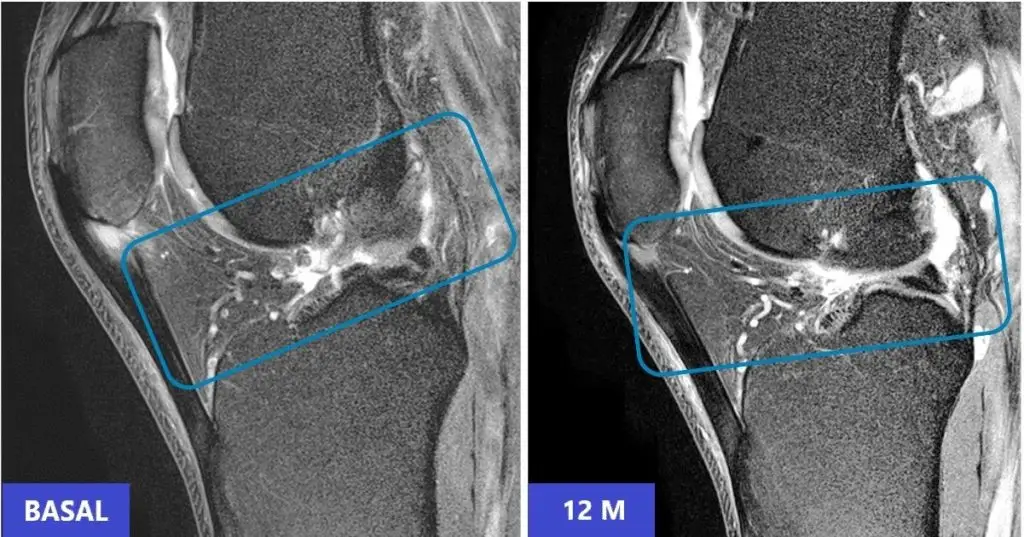
At 6 months, the investigators observed a clinically relevant difference: while both treatments reduced pain, only the CMC group showed signs of tendon tissue repair. In light of this finding, the study protocol allowed patients in the PRP group who had not improved structurally to also receive CMC in a second phase.
Main study results
| CMC (cultured mesenchymal cells) | PRP (platelet-rich plasma) | |
|---|---|---|
| Pain reduction at 6 months | Yes, with significant improvement | Yes, with significant improvement |
| Functional recovery (daily activity and sports) at 6 months | Yes, with significant improvement | Yes, with significant improvement |
| Structural tendon repair documented by imaging | Yes, only group with significant changes | No relevant changes observed |
| Imaging follow-up | MRI and specialized ultrasound at the same time points for both groups | Same follow-up protocol as the CMC group |
| Product classification | Advanced cell therapy medicinal product regulated by the AEMPS | Autologous biological procedure not classified as a medicinal product |
In summary, both treatments relieved pain to a similar extent, but only CMC were shown to structurally repair the tendon tissue at 6 and 12 months. PRP improved symptoms but did not produce visible repair of the damaged tendon on imaging. These findings were subsequently confirmed in a complementary analysis published in Biomedicines (2024), using a specialized ultrasound technique (UTC) that allows precise measurement of tendon tissue quality.
Clinical limitations of PRP in severe chronic tendinopathy
PRP is a generally well-tolerated procedure, with a useful anti-inflammatory effect in various clinical scenarios. However, in chronic tendinopathies with structural compromise it presents three relevant limitations: biological variability between centrifugation protocols and individual responses, with no universal standardization; absence of structural regeneration measurable by imaging when fiber separation ≥3 mm is present; and risk of injury progression, if the patient (with pain relieved) resumes physical activity with a structurally compromised tendon, which can increase the risk of complete tendon rupture.
When is an advanced cell therapy medicinal product with CMC indicated?
It is indicated in patients with refractory chronic patellar tendinopathy, particularly when:
- Previous conservative treatments (physical therapy, shockwave therapy, injections) have not provided lasting resolution.
- MRI shows tendon degeneration or structural tear with fiber separation.
- The patient requires full functional recovery (elite athletes or active patients).
The procedure is percutaneous, ultrasound-guided, under sedation. It takes approximately 45 minutes in the operating room. The patient walks out of the clinic and is able to travel between 24 and 48 hours after discharge.
Frequently asked questions
Does PRP contain stem cells?
No. PRP is a concentrate of platelets and growth factors obtained by centrifugation of the patient’s own blood. It does not contain stem cells and therefore cannot be considered a cell therapy or a stem cell treatment. Platelets are cellular fragments without a nucleus; they lack the self-renewal and differentiation capacity that defines a stem cell. This distinction matters because, in commercial communication, PRP is sometimes presented as a “stem cell” or “regenerative” treatment, a claim that is scientifically incorrect and recently addressed explicitly in the international scientific literature as a misconception that needs to be clarified.
CMC, by contrast, are living cells: mesenchymal cells extracted from the patient’s bone marrow and expanded in the laboratory under GMP cleanroom conditions, with demonstrated capacity to modulate inflammation and stimulate structural regeneration of the tissue.
How long does it take for a CMC treatment to work in the patellar tendon?
Response times vary from case to case, depending on the degree of tendon degeneration, the patient’s age, level of physical activity, and any associated conditions. As a general reference: pain improvement may be noticed from the first week; maximum functional recovery is reached around two months, and return to elite sports activity between 3 and 5 months. Full structural regeneration may take between 12 and 24 months, documented by MRI. Each patient is evaluated individually to determine their expected progression.
How is an advanced cell therapy medicinal product with CMC authorized in Spain?
CMC are a medicinal product regulated by the Spanish Agency of Medicines and Medical Devices (AEMPS) and by European regulations. Their authorization requires the prior completion of clinical trials that verify their safety, quality, efficacy, and manufacturing processes in a cleanroom under GMP standards. Only after rigorous clinical results are demonstrated do the product and its protocol of use receive formal authorization from the AEMPS. Throughout the entire treatment, each patient is registered with full traceability and remains under pharmacovigilance follow-up.
In patients with chronic patellar tendinopathy and a fiber tear visible on MRI, the evidence from the ITRT clinical trial shows that both treatments reduce pain, but only CMC were able to structurally repair the tendon tissue. Reducing pain is not the same as repairing the tendon.
Legal disclaimer
Advanced therapy medicinal products with cultured mesenchymal cells (CMC) require a medical prescription and individual authorization by the AEMPS. This content is for informational purposes only and does not substitute medical advice. The clinical results mentioned correspond to patients treated in the context of authorized clinical trials and are not extrapolatable to other patients.