An injury to the patellar tendon can severely impair joint function and complicate recovery. Treatment with cultured mesenchymal cells (CMC) enables the regeneration of damaged tissue. Additionally, this therapy helps restore the integrity of the patellar tendon, accelerates functional recovery, and improves the patient’s quality of life. Exploring the potential of cultured mesenchymal cells in treating patellar tendon injuries opens new avenues in sports and orthopedic medicine.
Function of the Patellar Tendon
The patellar tendon is a key structure for knee mobility and stability. Understanding its function and the importance of its regeneration is essential to effectively address its injuries:
- It connects the patella to the tibia, enabling the transmission of force necessary for leg extension.
- It facilitates essential movements such as walking, running, and jumping through its biomechanical function.
- It acts as a shock absorber by absorbing impact forces and protecting the knee joint.
When a patellar tendon injury occurs, patients face a number of challenges that traditional treatments often fail to address:
- Managing pain, inflammation, and loss of mobility while maintaining functional integrity.
- Preventing further injuries such as partial or complete tears caused by trauma, overuse, or degenerative processes like patellar tendinitis.
- Detecting tendon injuries early to avoid severe functional complications.
See also: Mesenchymal Cells in Tissue Regeneration: A Revolutionary Advancement in Regenerative Medicine
The Role of Cultured Mesenchymal Cells in Advanced Cell Therapy
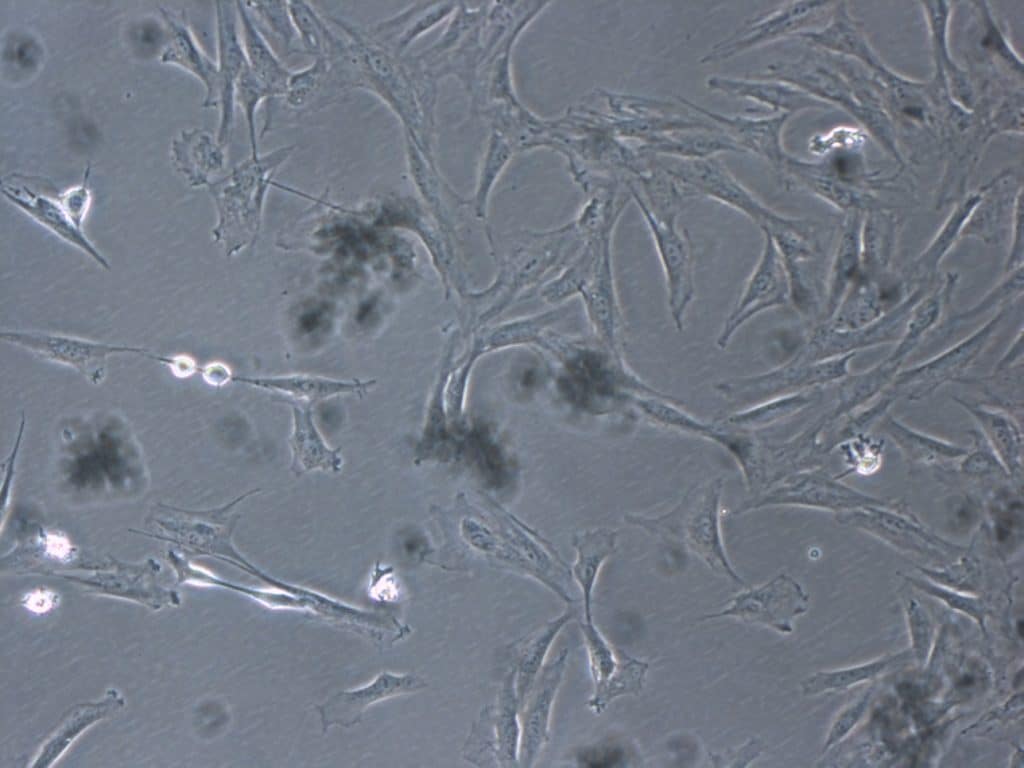
Understanding the potential of cultured mesenchymal cells (CMC) is critical to ensuring recovery in patients with patellar tendon injuries. Their regenerative capacity in tissues such as tendon, cartilage, and disc allows them to play a central role in repairing damaged tissue.
Bone marrow is a key source for obtaining mesenchymal cells for clinical use. These cells not only help replace damaged tissue, but also secrete growth factors and biochemical signals that modulate the cellular environment and promote tendon healing.
The use of cultured mesenchymal cells has marked a turning point in advanced therapy medicinal products by offering treatments that target the origin of tendon injuries, restore patellar tendon function, and objectively improve patients’ quality of life.
Clinical Evidence on Patellar Tendon Regeneration

The Institute of Tissue Regenerative Therapy (ITRT) is a pioneer in using cultured mesenchymal cells (CMC) to treat various musculoskeletal injuries. In the context of personalized treatments, ITRT has demonstrated clinically significant outcomes, with nearly 100% of treated patients experiencing some degree of functional recovery.
Key outcomes from ITRT in the treatment of patellar tendon ruptures and general tendon injuries include:
- Over 300 tendons regenerated using CMC.
- Over 70 patellar tendons regenerated through CMC therapy.
- A clinical trial was conducted to evaluate tendon regeneration using cultured mesenchymal cells. To date, ITRT is the only center worldwide to have demonstrated clinical evidence of tendon regeneration and healing in patients, with results published in a high-impact scientific journal.
The American Journal of Sports Medicine highlighted the results of intratendinous and peritendinous application of bone marrow-derived mesenchymal stem cells, which showed exceptional recovery in athletes with patellar tendinopathy.

Twenty patients aged 18 to 48, with varying degrees of pain lasting over four months and who had not responded to conventional non-surgical treatments, were included in the study.
The patients were divided into two groups and treated with 20 × 10⁶ bone marrow-derived mesenchymal stem cells (BM-MSC) and leukocyte-poor platelet-rich plasma (Lp-PRP). Six months after treatment, patients showed improvement in tendon structure restoration, with many resuming sports activities just two months after treatment.
Traditional Treatments vs. Cultured Mesenchymal Cell Therapy
Comparing conventional treatments with cultured mesenchymal cell (CMC) therapy helps identify more effective approaches to promote patellar tendon regeneration:
| Traditional Treatments for the Patellar Tendon | Cultured Mesenchymal Cell Therapy |
|---|---|
| Focus on symptom relief through physical therapy, medications, or surgery. | Regenerate the patellar tendon structurally and functionally. |
| Reduce pain and inflammation without repairing internal damage. | Promote cellular differentiation and secrete growth factors that restore the tendon. |
| Provide temporary relief with a risk of recurrence. | Provide long-term recovery and reduce the risk of reinjury. |
| Do not address the biological cause of tendon damage. | Target the root cause by regenerating the damaged tissue. |
| Success depends heavily on injury severity and adherence to rehabilitation. | Partially independent of rehabilitation compliance due to biological regeneration. |
| Surgery as the last resort, with partial recovery risks. | Avoid surgery in many cases through advanced tissue repair. |
Legal and Regulatory Considerations
Regulating the use of mesenchymal cells in regenerative medicine is essential to ensure treatment safety, efficacy, and traceability in Spain. Following the guidelines set by the Spanish Agency of Medicines and Medical Devices (AEMPS) ensures that cell therapies are developed and applied in a controlled and legally compliant manner.
AEMPS authorization for clinical trials is a mandatory step to demonstrate the efficacy, safety, proper dosage, administration route, and adverse effect profile of any advanced therapy medicinal product before it is used in clinical practice.
Ethical principles must also be upheld when obtaining cells, ensuring informed donor consent and proper handling, processing, and preservation of cultured mesenchymal cells. These measures are vital to guarantee therapeutic quality, viability, and reproducibility of the treatment.
Complying with this legal and ethical framework is not only mandatory but essential to advancing safe, scientifically validated, and reliable personalized therapies for patients.

What Is the Next Step for Patients with Patellar Tendon Injuries?
The first step is a precise diagnosis using clinical evaluation and MRI imaging, followed by assessing the extent of the injury to determine the most appropriate treatment.
In severe or chronic cases, evaluating mesenchymal cell therapy as the most effective option is essential. Consultation with ITRT will help assess the feasibility of treatment with cultured mesenchymal cells. The process begins with harvesting, processing, and administering the cells to the injury site, initiating the therapeutic regeneration process.
Ongoing clinical follow-up and allowing sufficient time for biological regeneration contribute to a more complete recovery and significant functional improvement of the patellar tendon.