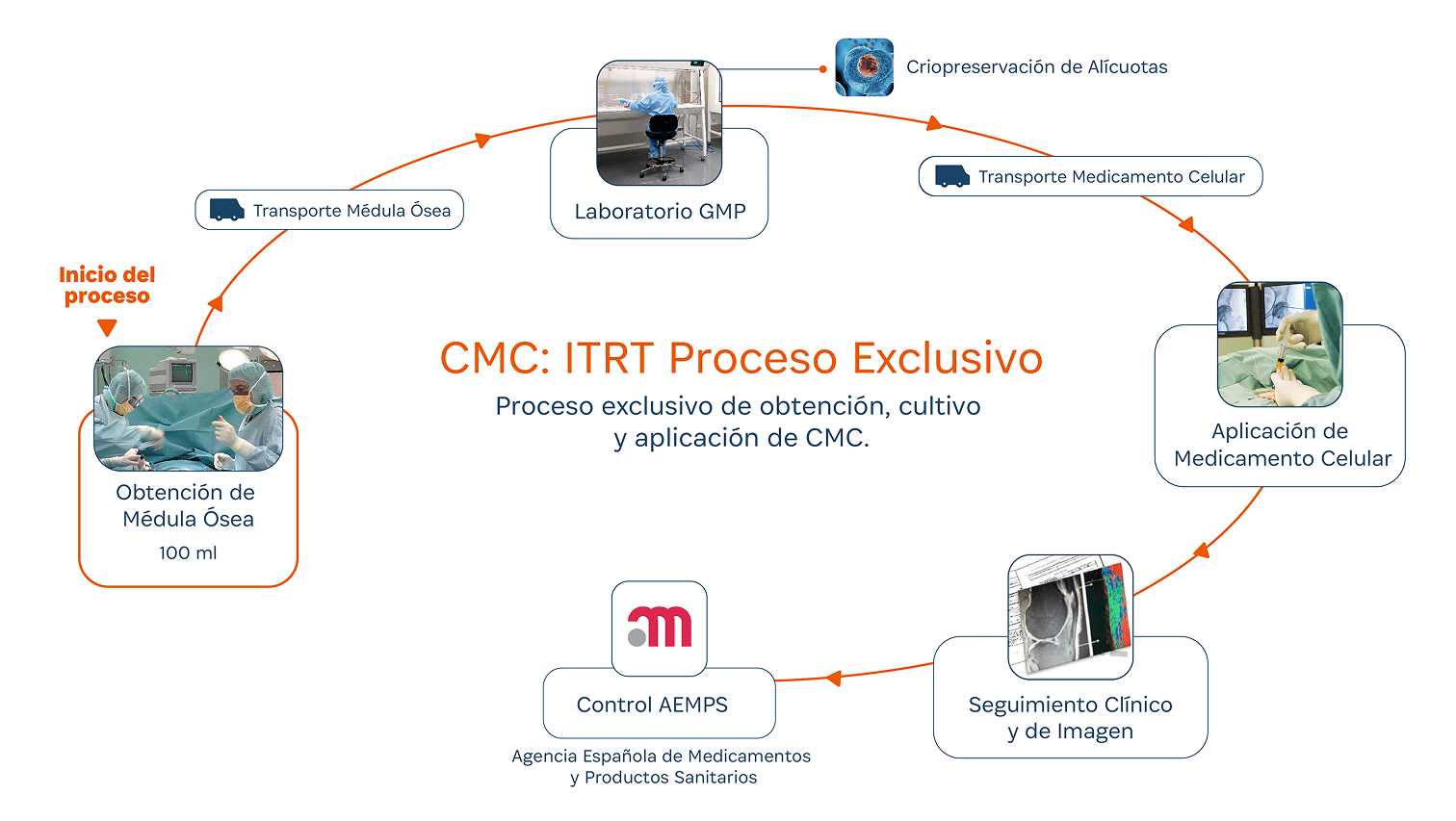
What is required to carry out a treatment with an advanced therapy medicinal product?
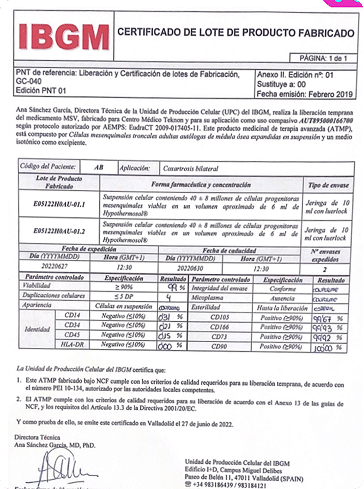
Through an authorized clinical trial, in accordance with current legislation. The European SoHO (Substances of Human Origin) regulation establishes that conducting a clinical trial is mandatory in order to develop an advanced therapy medicinal product.
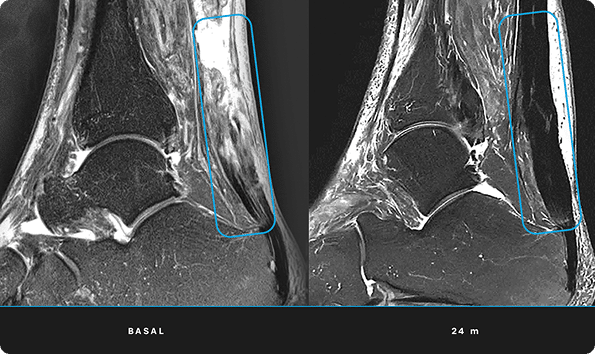
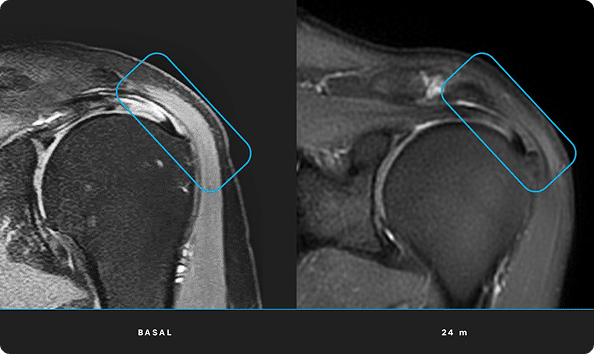
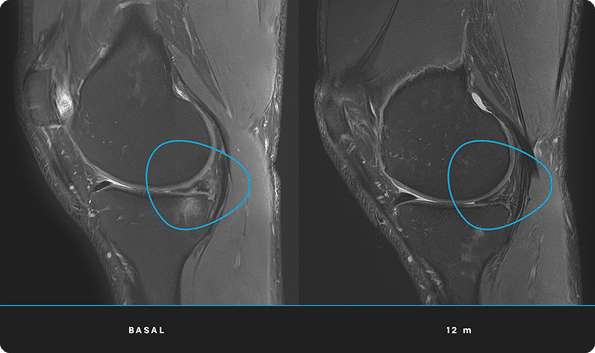
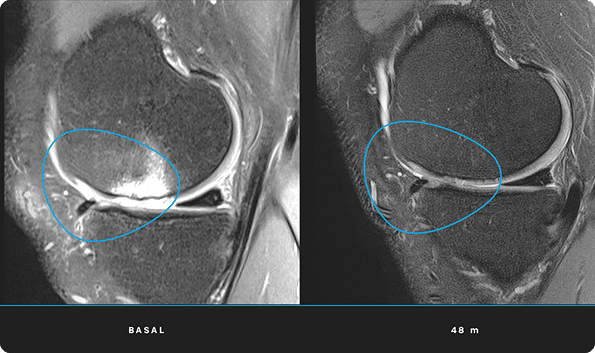
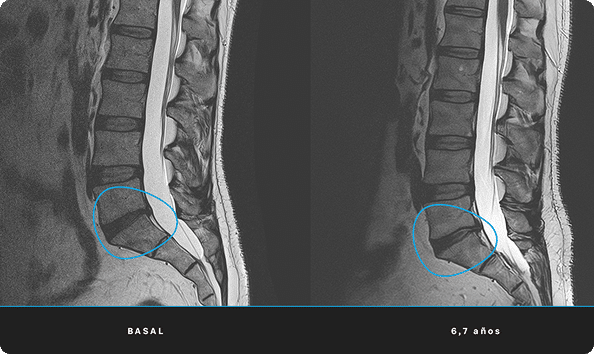
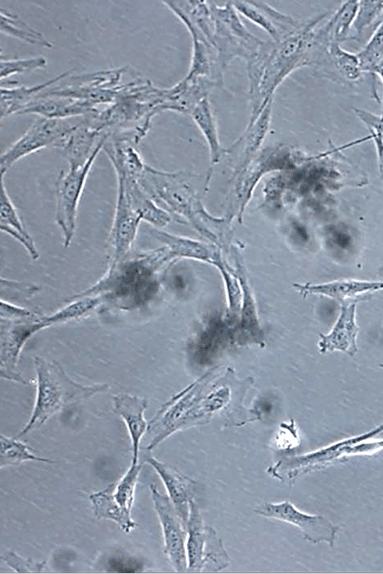
At ITRT, we have demonstrated the feasibility and safety of our therapies through clinical trials regulated and authorized by the Spanish Agency of Medicines and Medical Devices (AEMPS). Once the clinical trial has been completed, the AEMPS authorizes the use of the medicinal product in specific patients, allowing us to offer personalized treatments with proven clinical efficacy and regenerative capacity.