La Vanguardia: regenerative therapies restore Achilles tendon rupture without surgery
The major paradigm shift in the treatment of this injury has been the introduction of cultured mesenchymal cells, administered percutaneously under deep sedation.

The major paradigm shift in the treatment of this injury has been the introduction of cultured mesenchymal cells, administered percutaneously under deep sedation.
Before undergoing any procedure involving mesenchymal cells, there is one question that should be answerable with documentation: how many cells will be administered, and how has that number been verified? If no concrete answer exists, the procedure is not classified as a regulated medicinal product. The cellular dose is the technical and regulatory criterion that marks that difference. In this article, we explain how it is defined, how it is certified, and why it is decisive in indications such as tendons, cartilage, and lumbar disc disease, from the clinical and scientific perspective of ITRT.
Current research analyzes how to intervene in damaged structures in cases of knee osteoarthritis through advanced therapy medicinal products with cultured mesenchymal cells (CMC), evaluated in authorized clinical trials. Addressing how to regenerate cartilage requires distinguishing between non-regulated procedures and medicines developed under European regulatory standards.

The production of cultured mesenchymal cells (CMC) in a GMP cleanroom transforms a biological material into an advanced therapy medicinal product under quality control and regulatory supervision.

European and Spanish legislation establishes that only advanced therapy medicinal products authorized by the AEMPS can be considered regenerative cellular treatments. This article explains how to distinguish regulated medical interventions from unauthorized practices that use human cells without clinical evidence or legal approval, offering a rigorous analysis within the framework of the SoHO Regulation and ITRT’s clinical experience.
Cultured Mesenchymal Cells (CMC) are applied within AEMPS-authorized clinical protocols to support structural and functional recovery of the patellar tendon in selected cases of chronic tendinopathy. Their use follows a regulated, personalized approach with GMP manufacturing and medical supervision.

Dr Robert Soler presented results from advanced therapy medicinal products authorised by AEMPS to a world-class international panel, positioning ITRT as a European reference in musculoskeletal regeneration

Chronic patellar tendinopathy can be regenerated without surgery through advanced cell therapy. ITRT leads this approach with clinical evidence, regulated manufacturing, and over 314 doses administered using Cultured Mesenchymal Stem Cells (CMC).
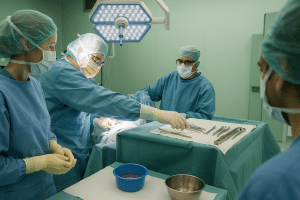
At ITRT, we address pseudoarthrosis with a rigorous clinical approach, combining expert diagnosis, scientific evidence, and advanced cellular therapies. We apply cultured mesenchymal cells (CMC) as an advanced therapy medicinal product, developed under GMP conditions and individually authorized by the AEMPS. Our therapeutic model ensures traceability, safety, and regulated clinical follow-up, focused on restoring bone function in non-healing fractures. Because each case requires precision, personalization, and real medicine.
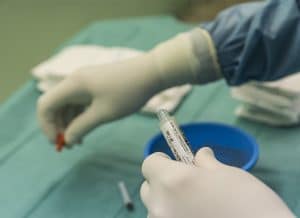
ITRT’s clinical trials with cultured mesenchymal cells (CMC) have shown excellent results in the treatment of tissue injuries.

Scientific and regulatory information on the treatment of tendinopathies with advanced cell therapy products.
At ITRT, we offer a clear guide before starting any cell-based treatment.
Don’t be misled—know the key questions to ask and ensure you’re receiving an authorized cell-based medicine with clinical evidence and regulated follow-up.

The Use of Cultured Mesenchymal Cells (CMC) Is Essential in Regenerating the Achilles Tendon Across Various Types of Injuries.

The supraspinatus tendon supports joint mobility and provides stability. When injured, this critical function is compromised, often accompanied by chronic pain and daily activity limitations.

Chronic low back pain is often associated with factors such as mechanical overload, disc aging, sedentary behavior, or long-term postural imbalances. These factors contribute to the progressive degeneration of the intervertebral discs in the lumbar region, a condition also known as black disc disease.

Regenerating the patellar tendon through an advanced therapy medicinal product represents a significant breakthrough in personalized medicine. This tendon, which connects the patella to the tibia, plays a crucial role in knee mobility and stability.

The case of Pau Gasol, one of the most prominent figures in Spanish sports and a former NBA player for 18 seasons, is particularly significant. Gasol was one of the

Over the past decade, mesenchymal cells have emerged as one of the most promising solutions in regenerative medicine, particularly for tendon regeneration without surgery, delivering scientifically proven results. These cultured

On March 21–22, the Grand Hotel Dino in Baveno, Italy, hosted Joint Challenge 2025—an international forum that brought together leading experts in orthopaedics, rheumatology, sports medicine and rehabilitation to explore

On August 7, Dr. Robert Soler Rich, Medical Director of the ITRT, gave the presentation “Treatment with cultured stem cells from bone marrow in diseases of the Locomotor System” in

Data from the clinical trial, which has just been published in the prestigious American Journal of Sports Medicine, provides new scientific evidence and opens up a therapeutic option, since the
Advanced cell therapy medicinal products based on Cultured Mesenchymal Cells (CMC) are subject to medical prescription and require individual authorization from the Spanish Agency of Medicines and Medical Devices (AEMPS). The information presented here is for educational purposes only and does not replace medical consultation or professional healthcare advice.
© Copyright 2025 ITRT – All rights reserved. Website owned by ITRT.
